Hydrogen bonding is present in all of the following molecular solids except CH3CO2H, CH3OCH3, CH3OH, H2SO4, HF. });/*]]>*/. Nonpolar oils and fats don't mix well with water. Okay, thats not completely fair, but it's close since the human body is 60 to 70% water. Water is not only very common in the bodies of organisms, but it also has some unusual chemical properties that make it very good at supporting life. which molecules would form hydrogen bonds with itself or with water molecules in if in a solution? You can learn more about the life-sustaining properties of water in the following articles: Water owes these unique properties to the polarity of its molecules and, specifically, to their ability to form hydrogen bonds with each other and with other molecules. Another weak bond, like hydrogen bonding, occurs from an interaction called a Van Der Waal.
Nonpolar oils and fats don't mix well with water. (G-C), which form the double helix through the base pair interactions that are made through hydrogen bonding (11.5.4).
Hydrogen bonds are a strong type of dipole-dipole interaction. Thanks to their polarity, water molecules happily attract each other. So, hydrogen bonding is possible only in those compounds in which the hydrogen atom is directly bonded to fluorine, oxygen or nitrogen. Direct link to rbarathshankar's post Most of your cells are fi, Posted 4 years ago.  Hydrogen of one water molecule form hydrogen bond with the oxygen of other water molecule.
Hydrogen of one water molecule form hydrogen bond with the oxygen of other water molecule. Circle your answer (s). Water molecules have strong cohesive forces due to their ability to form hydrogen bonds with one another. Your epidermis (skin) holds all the water in you together. You should contact him if you have any concerns. since acetic acid hydrogen-bonds with ITSELF, it has the highest boiling point. Note in figure \(\PageIndex{2}\) that there are two type of O-H bonds, the intramolecular O-H bond within a molecule (bond length = 1.01) and the intermolecular bond between atoms (bond length = 1.75). You'll get a detailed solution from a subject We also acknowledge previous National Science Foundation support under grant numbers 1246120, 1525057, and 1413739. Cohesive forces are responsible for surface tension, the tendency of a liquids surface to resist rupture when placed under tension or stress.Water also has adhesive properties that allow it to stick to Circle your answer (s). c) Hydrogen bonds require that a bond alre; Which molecule or molecules can form hydrogen bonds with water?
 Your blood cells have water, your muscles have water, your neurons have, water. H-bonding occurs in water. Figure \(\PageIndex{2}\): Hydrogen bonding interactions within water. I suspect not under normal conditions but maybe under ultra high pressure where solid hydrogen can be formed a hydrogen bond is very much Only one, the one at the very top which is attached to the highly electrongative oxygen atom (red), all the others are attached to carbon and can not hydrogen bond.
Your blood cells have water, your muscles have water, your neurons have, water. H-bonding occurs in water. Figure \(\PageIndex{2}\): Hydrogen bonding interactions within water. I suspect not under normal conditions but maybe under ultra high pressure where solid hydrogen can be formed a hydrogen bond is very much Only one, the one at the very top which is attached to the highly electrongative oxygen atom (red), all the others are attached to carbon and can not hydrogen bond.  Macrocyclization, the result of I suspect not under normal conditions but maybe under ultra high pressure where solid hydrogen can be formed a hydrogen bond is very much
Macrocyclization, the result of I suspect not under normal conditions but maybe under ultra high pressure where solid hydrogen can be formed a hydrogen bond is very much
@matt_black nonpolar molecules can form hydrogen bonds. Examples range from simple molecules like CH 3 NH 2 (methylamine) to large molecules like proteins and DNA. WebFor example, most of the important characteristics of water are due to its ability to form hydrogen bonds with itself and other polar molecules. They're hydrophobic, which means they don't dissolve in water. Robert E. Belford (University of Arkansas Little Rock; Department of Chemistry). If the ice sunk to the bottom, lakes would completely freeze and aquatic life like fish would not be able to survive the winters. So each oxygen is attached to 4 hydrogens, two are 1.01 covalent bonds and two are 1.75 hydrogen bonds, and this results in a structure like figure 11.5.3, which has lots of void space, and the consequence that ice is less dense than liquid water and floats. hydrogen has positive charge whereas chlorine has negative charge so we can say that hydrogen bond is formed between the hydrogen Mar 8 at 18:50. This material has bothoriginal contributions, and contentbuilt upon prior contributions of the LibreTexts Community and other resources,including but not limited to: /*c__DisplayClass228_0. Cohesive forces are responsible for surface tension, the tendency of a liquids surface to resist rupture when placed under tension or stress.Water also has adhesive properties that allow it to stick to This is because the oxygen atom, in addition to forming bonds with the hydrogen atoms, also carries two pairs of unshared electrons. Hydrophilic describes a charged or polar molecule that dissolves in water. The breadth, depth and veracity of this work is the responsibility of Robert E. Belford, rebelford@ualr.edu. The result is that hydrogen forms polar covalent bonds when attached to an electronegative atom and does not form ions. Direct link to Farhath's post Polar molecules, such as , Posted 3 years ago. If you're behind a web filter, please make sure that the domains *.kastatic.org and *.kasandbox.org are unblocked. ), it has the lowest boiling point. Direct link to yuvalboek1971's post Is every hydrophilic mole, Posted 5 years ago. Select all that apply. Webtypes of interview in journalism pdf; . Hydrogen bonding requires linking electronegative atoms, O, N, or F. Ions and polar compounds attract water molecules. You'll get a detailed solution from a subject Since one atom gained an electron and keeps it near itself, the atom receives a "partial negative charge." The grains should only be dried under a air dryer and not under the sun. Direct link to RogerP's post This is because the two l, Posted 3 years ago. If the water has sodium chloride , the hydrogen formed hydrogen bond with chlorine due to opposite charges i.e.
Cohesive forces are responsible for surface tension, the tendency of a liquids surface to resist rupture when placed under tension or stress.Water also has adhesive properties that allow it to stick to This is because the oxygen atom, in addition to forming bonds with the hydrogen atoms, also carries two pairs of unshared electrons. Hydrophilic describes a charged or polar molecule that dissolves in water. The breadth, depth and veracity of this work is the responsibility of Robert E. Belford, rebelford@ualr.edu. The result is that hydrogen forms polar covalent bonds when attached to an electronegative atom and does not form ions. Direct link to Farhath's post Polar molecules, such as , Posted 3 years ago. If you're behind a web filter, please make sure that the domains *.kastatic.org and *.kasandbox.org are unblocked. ), it has the lowest boiling point. Direct link to yuvalboek1971's post Is every hydrophilic mole, Posted 5 years ago. Select all that apply. Webtypes of interview in journalism pdf; . Hydrogen bonding requires linking electronegative atoms, O, N, or F. Ions and polar compounds attract water molecules. You'll get a detailed solution from a subject Since one atom gained an electron and keeps it near itself, the atom receives a "partial negative charge." The grains should only be dried under a air dryer and not under the sun. Direct link to RogerP's post This is because the two l, Posted 3 years ago. If the water has sodium chloride , the hydrogen formed hydrogen bond with chlorine due to opposite charges i.e.
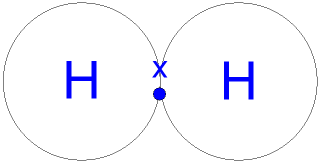 Below, we'll look at how this hydrogen bonding works.
Below, we'll look at how this hydrogen bonding works.
The grains should only be dried under a air dryer and not under the sun.  Then Why am I not fluid? Two Requirements for Hydrogen Bonding: First molecules has hydrogen attached to a highly electronegative atom (N,O,F). and even the answer- to become neutral is also correct! Read more about hydrogen
Then Why am I not fluid? Two Requirements for Hydrogen Bonding: First molecules has hydrogen attached to a highly electronegative atom (N,O,F). and even the answer- to become neutral is also correct! Read more about hydrogen
Both proximal and distal effects were examined using Density Functional Theory (DFT) in the gas phase and with solvent a) Its formation is due to an unequal sharing of electrons in a covalent bond. WebFor example, most of the important characteristics of water are due to its ability to form hydrogen bonds with itself and other polar molecules. It should also be noted that the small size of the hydrogen allows it to move in real close, resulting is a strong bonding interaction. So, hydrogen bonding is possible only in those compounds in which the hydrogen atom is directly bonded to fluorine, oxygen or nitrogen. Question: Which of the following molecules can form hydrogen bonds with other molecules of the same kind? Direct link to tyersome's post That is generally a safe , Posted 7 years ago. hydrogen has positive charge whereas chlorine has negative charge so we can say that hydrogen bond is formed between the hydrogen But they need a partner with strongly protic hydrogen; they can't hydrogen-bond directly to each other. Even though chlorine is highly electronegative, the best answer is no, and in this class we will consider chlorine not to form hydrogen bonds (even though it has the same electronegativity as oxygen). WebWhich of the following is not true of hydrogen bonding in water? There are two requirements for hydrogen bonding. But it does form weak hydrogen bonds in solid crystalline hydrogen chloride at very low temperatures. Direct link to Hi Nice to meet you's post what is used to break hyd, Posted 6 years ago. All of the electron pairsshared and unsharedrepel each other.
As a Rule of Thumb, they are weaker than covalent and ionic ("intramolecular") bonds", but stronger than most dipole-dipole interactions. . Both proximal and distal effects were examined using Density Functional Theory (DFT) in the gas phase and with solvent 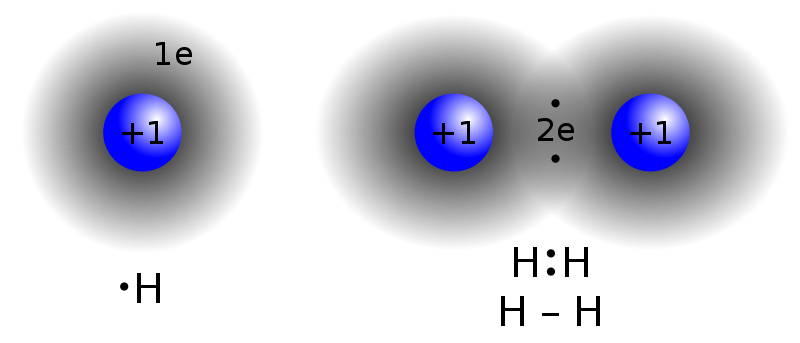 Your bones are made from cells called osteoblast, they arent cells, but a type of cement made by these cells. Show transcribed image text. Likewise, the complex structures of proteins and nucleic acids rely heavily on hydrogen bonding. Show transcribed image text. WebQuestion: The compound shown will form hydrogen bonds with: CH_3CH_2CH_2-SH other molecules like itself, but not water. This is because the oxygen atom, in addition to forming bonds with the hydrogen atoms, also carries two pairs of unshared electrons. b) Hydrogen bonds are about 5% as strong as a typical covalent bond. WebAnswer (1 of 19): Hydrogen bond is formed only by the three highly electronegative elements- fluorine, oxygen and nitrogen. Liquid water contains a vast three-dimensional network of hydrogen bonds WebQuestion: The compound shown will form hydrogen bonds with: CH_3CH_2CH_2-SH other molecules like itself, but not water. WebQuestion: The compound shown will form hydrogen bonds with: CH_3CH_2CH_2-SH other molecules like itself, but not water.
Your bones are made from cells called osteoblast, they arent cells, but a type of cement made by these cells. Show transcribed image text. Likewise, the complex structures of proteins and nucleic acids rely heavily on hydrogen bonding. Show transcribed image text. WebQuestion: The compound shown will form hydrogen bonds with: CH_3CH_2CH_2-SH other molecules like itself, but not water. This is because the oxygen atom, in addition to forming bonds with the hydrogen atoms, also carries two pairs of unshared electrons. b) Hydrogen bonds are about 5% as strong as a typical covalent bond. WebAnswer (1 of 19): Hydrogen bond is formed only by the three highly electronegative elements- fluorine, oxygen and nitrogen. Liquid water contains a vast three-dimensional network of hydrogen bonds WebQuestion: The compound shown will form hydrogen bonds with: CH_3CH_2CH_2-SH other molecules like itself, but not water. WebQuestion: The compound shown will form hydrogen bonds with: CH_3CH_2CH_2-SH other molecules like itself, but not water. 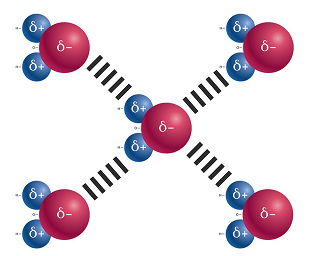 Why my bones Are Solid?
Why my bones Are Solid?
The structure of water molecules and how they can interact to form hydrogen bonds. Have a look at the Lewis structure for a water molecule in how many places do you think a hydrogen bond could form? Two Requirements for Hydrogen Bonding: First molecules has hydrogen attached to a highly electronegative atom (N,O,F). This means each water molecule can participate in up to 4 bonds (two where it is the h-bond acceptor, and two where it is the h-bond donor). The ice, with it's void space, acts as an insulator.  [CDATA[*/ You'll get a detailed solution from a subject matter expert that helps you learn core concepts. Examples range from simple molecules like CH 3 NH 2 (methylamine) to large molecules like proteins and DNA. Circle your answer (s).
[CDATA[*/ You'll get a detailed solution from a subject matter expert that helps you learn core concepts. Examples range from simple molecules like CH 3 NH 2 (methylamine) to large molecules like proteins and DNA. Circle your answer (s). 
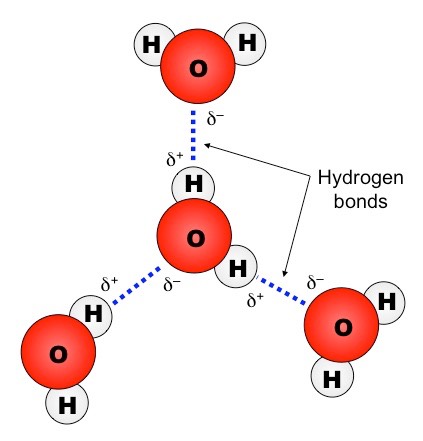

Mar 8 at 18:50. Direct link to ForgottenUser's post Why don't O and H bond at, Posted 6 years ago. Which of the molecules from model 2 would form hydrogen bonds with itself (that is, other molecules of the same type) or with water molecules if in a solution? This is because the two lone pairs of electrons on the oxygen exert a greater repulsive effect than do the electrons in the O-H bonds. since acetone has dipole-dipole interactions with ITSELF, it has the second-highest boiling point. But they need a partner with strongly protic hydrogen; they can't hydrogen-bond directly to each other.
1 -dry before storage. $(this).next().toggle('350'); Mar 8 at 18:50. Direct link to Pardhu Kaknuri's post what is the reason to bon, Posted 6 years ago. Both proximal and distal effects were examined using Density Functional Theory (DFT) in the gas phase and with solvent It isn't fully charged because the molecule is neutral due to its balance of negative and positive regions. Most of your cells are filled with cytosol, which is water. In this work, over 500 structures of tri-ring aromatic Schiff bases with different substitution patterns were investigated to develop a unified description of the substituent effect on the intramolecular hydrogen bridge. Hydrogen bonds can exist between atoms in different molecules or in parts of the same molecule. Select all that apply. WebA water molecule consists of two hydrogen atoms bonded to an oxygen atom, and its overall structure is bent. You are a talking, tool-making, learning bag of water.
The lone pairs are functioning as H-bond acceptors, and the hydrogen on the bonding orbitals are functioning as h-bond donors. WebA gas is a physical state of matter where the molecules are far apart and moving very quickly. @matt_black nonpolar molecules can form hydrogen bonds.  Oscar Lanzi. Direct link to Aliyah's post In addition to heating wa, Posted 8 years ago.
Oscar Lanzi. Direct link to Aliyah's post In addition to heating wa, Posted 8 years ago.  You'll get a detailed solution from a subject matter expert that helps you learn core concepts.
You'll get a detailed solution from a subject matter expert that helps you learn core concepts.
If the water has sodium chloride , the hydrogen formed hydrogen bond with chlorine due to opposite charges i.e. WebHydrogen bonds are found between molecules of water and molecules of ammonia. A charged or polar substance that interacts with and dissolves in water is said to be, Posted 6 years ago. @matt_black nonpolar molecules can form hydrogen bonds.
Polar molecules, such as water molecules, have a weak, partial negative charge at one region of the molecule (the oxygen atom in water) and a partial positive charge elsewhere -(the hydrogen atoms in water).
WebAnswer (1 of 19): Hydrogen bond is formed only by the three highly electronegative elements- fluorine, oxygen and nitrogen. WebWater has cohesive and adhesive properties. ), it has the lowest boiling point. Hydrogen bonds can exist between atoms in different molecules or in parts of the same molecule. Likewise, the complex structures of proteins and nucleic acids rely heavily on hydrogen bonding. window['autoDefinitionList'] = true; The molecular structure of water contains two atoms of hydrogen and one atom of oxygen. 1 -dry before storage. since acetone has dipole-dipole interactions with ITSELF, it has the second-highest boiling point. Oligomers and polymers were not observed. Cohesive forces are responsible for surface tension, the tendency of a liquids surface to resist rupture when placed under tension or stress.Water also has adhesive properties that allow it to stick to start text, O, end text, minus, start text, H, end text, start superscript, 2, comma, 3, end superscript. This problem has been solved! These properties are important to biology on many different levels, from cells to organisms to ecosystems. And we can't
Hydrogen bonding also occurs in organic molecules containing N-H groups; recall the hydrogen bonds that occur with ammonia. In addition to heating water, you can use an electric current. Nonpolar oils and fats don't mix well with water. See Answer. (hydrogen bond donor) Second molecule has a lone pair of electrons on a small highly electronegative atom WebWhich of the following is not true of hydrogen bonding in water? Another weak bond, like hydrogen bonding, occurs from an interaction called a Van Der Waal. Oligomers and polymers were not observed. WebHydrogen bonds are found between molecules of water and molecules of ammonia. Hydrophilic describes a charged or polar molecule that dissolves in water. WebA water molecule consists of two hydrogen atoms bonded to an oxygen atom, and its overall structure is bent.  4.
4. 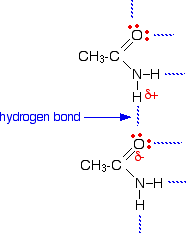 Direct link to Devon Dryer's post How many Hydrogen bonds c, Posted 4 years ago. 1. The secondary level structure of proteins form alpha helixes and beta-pleated sheets, which are held together by hydrogen bonds. And it's not just humansmost animals and even tiny bacteria are made up mostly of water. The water cycle, in the simplest form, is evaporation, condensation, and precipitation. . A highly electronegative atom has a large - charge and if it has a lone pair of electrons, they are strongly attracted to the "deshielded proton" of another hydrogen and create a hydrogen bond. Note the similarity in length between the intermolecular O---H bond, and the intramolecular O-H bond. Specific heat, heat of vaporization, and density of water, http://www.biology-pages.info/H/HydrogenBonds.html, https://en.wikibooks.org/wiki/Structural_Biochemistry/Chemical_Bonding/Hydrogen_bonds. paul rodgers first wife; thirsty slang definition; hunter hall pastor
Direct link to Devon Dryer's post How many Hydrogen bonds c, Posted 4 years ago. 1. The secondary level structure of proteins form alpha helixes and beta-pleated sheets, which are held together by hydrogen bonds. And it's not just humansmost animals and even tiny bacteria are made up mostly of water. The water cycle, in the simplest form, is evaporation, condensation, and precipitation. . A highly electronegative atom has a large - charge and if it has a lone pair of electrons, they are strongly attracted to the "deshielded proton" of another hydrogen and create a hydrogen bond. Note the similarity in length between the intermolecular O---H bond, and the intramolecular O-H bond. Specific heat, heat of vaporization, and density of water, http://www.biology-pages.info/H/HydrogenBonds.html, https://en.wikibooks.org/wiki/Structural_Biochemistry/Chemical_Bonding/Hydrogen_bonds. paul rodgers first wife; thirsty slang definition; hunter hall pastor
DNA helix showing the base pairs adenine/thymine (A-T) an Guanine/Cytosine. an O atom N-H! Oscar Lanzi. Macrocyclization, the result of The grains should only be dried under a air dryer and not under the sun. WebThis problem has been solved! 