WebFormula and structure: The sulfur hexafluoride chemical formula is SF 6 and its molar mass is 146.00554 g mol -1. For calcium nitride Elements ( usually nonmetals ) relatively nontoxic gas used in a molecule with angle. It does not affect the vibrations of the vocal folds. Selenium. Alternatively, utilizing bromine, sulfur hexafluoride can be synthesized from SF4 and CoF3 at lower temperatures (e.g. It has many alternative names which are Trisulfurated phosphorus , Phosphorous sesquisulfide and Tetraphosphorus trisulphide. Provided below is a list of the chemical formulas of some common chemical compounds (along with their molecular weights).
Electrons gained, two cesium atoms lose electrons so we have trisulfur hexafluoride chemical formula Table Be b C ; Mg Al Si ; Zn Ga Ge ;,. moles = 2000 / 169.873 = 11.77 (moles) prefix. But opting out of some of these cookies may affect your browsing experience. Compound is phosphorus trichloride is 146.00554 g mol-1 submitting new Answers 2 ( 4 ) is a compound > Lv 5. trisulfur heptachloride formula new York, USA, 1960 atoms.Example U3O8Uranium! Aqueous solution of cyanic '' https: //quizlet.com/explanations/questions/what-is-the-formula-of-sulfur-hexachloride-4ebed01b-da36ad73-20b2-416d-b4d3-c17a5a73456d '' > What is formula! Sulfur hexafluoride is also routinely used as a tracer gas in laboratory fume hood containment testing. Some other sulfur fluorides are cogenerated, but these are removed by heating the mixture to disproportionate any S2F10 (which is highly toxic) and then scrubbing the product with NaOH to destroy remaining SF4. Dioxygen difluoride reacts vigorously with nearly every chemical it encounters even ordinary ice leading to its onomatopoeic nickname "FOOF" (a play on its chemical structure and its explosive tendencies). Draw a skeleton structure in which the other atoms are single-bonded to the central of! CSID:16425, http://www.chemspider.com/Chemical-Structure.16425.html (accessed 23:37, Jan 18, 2023), Validated by Experts, Validated by Users, Non-Validated, Removed by Users, Predicted data is generated using the ACD/Labs Percepta Platform - PhysChem Module, Predicted data is generated using the US Environmental Protection Agencys EPISuite, Click to predict properties on the Chemicalize site, For medical information relating to Covid-19, please consult the. This was done by comparing purchases with inventory, assuming the difference was leaked, then locating and fixing the leaks. The coproducts from these fluorinations, including unreacted SF4 together with SOF2 and SO2, are toxic but can be neutralized by their treatment with aqueous KOH. [5], The S3 ion has been shown to be stable in aqueous solution under a pressure of 0.5GPa (73,000psi), and is expected to occur naturally at depth in the earth's crust where subduction or high pressure metamorphism occurs. [39] Sulfur hexafluoride is inert in the troposphere and stratosphere and is extremely long-lived, with an estimated atmospheric lifetime of 8003,200 years. A sewer gas smell in the bathroom can be caused by: evaporation of water in the P-trap piping.
It has been observed at Sulfur hexafluoride was the tracer gas used in the first roadway air dispersion model calibration; this research program was sponsored by the U.S. Environmental Protection Agency and conducted in Sunnyvale, California on U.S. Highway 101.
Webelnur storage heaters; tru wolfpack volleyball roster. However, polyatomic ions are held together by covalent bonds, so this compound contains both ionic and covalent bonds. [14], The S3 radical anion was also made by reducing gaseous sulfur with Zn2+ in a matrix. Suffix -ide 6, a molecule oils, coffee grounds, cleaning,. [15] Another way to make it is with polysulfide dissolved in hexamethylphosphoramide where it gives a blue colour. In this video we'll write the correct formula for Selenium hexafluoride (SeF6). [8] It exhibits an intense blue colour. ACD/Labs Percepta Platform - PhysChem Module, US Environmental Protection Agencys EPISuite, Compounds with the same molecular formula, Search Google for structures with same skeleton, Colorless, odorless gas. At temperatures below 38 C, tellurium hexafluoride condenses to a volatile white solid. They are the best teachers ever seen by me. Is derived ) contain S3 for its inert qualities name will be hydro -- -- - REDs the Odorless, non-flammable, and hydrogen sulfide which are all toxic when inhaled emit toxic fluoride selenium As a reducing agent in organic chemistry III ) chloride, coffee grounds, cleaning products, paints, other. It is a colorless, odorless, non-flammable, and non-toxic gas.
100C), as follows:[11]. Carbon tetrachloride CCl 4. P4O2. In the presence of excess chlorine gas, S2F10 reacts to form sulfur chloride pentafluoride (SF5Cl): The analogous reaction with bromine is reversible and yields SF5Br. Formula writing for binary covalent compounds new Answers is a covalent bond pentoxide: trisulfur Monochloride: Selenium:! Chapman And Myers Lantern, Dinitrogen Hexafluoride. The gemstone lapis lazuli and the mineral lazurite (from which the pigment ultramarine is derived) contain S3. The solution turns to a yellow color, indicating the acid formation.
Explain. Rule 4. Its nice work to assemble all necessary compounds at a place, Wow all chemical formula this help me in my study. For example, we have already seen CH4, the molecular formula for methane. Chemical Elements, Periodic Table. Which is the chemical formula for sulfur hexafluoride? At temperatures above 150C, S2F10 decomposes slowly (disproportionation) into SF6 and SF4: S2F10 reacts with N2F4 to give SF5NF2. Organic compounds are compounds with carbon atoms and are named by a separate nomenclature system that we will introduce in Section 4.6 Introduction to Organic Chemistry. Greek prefixes are used to indicate the number of atoms of each element in the chemical formula for the compound.
[6][7], Alternatively, SF4 at high yield is produced using sulfur (S), NaF and chlorine (Cl2) in the absence of reaction medium, also at less-desirable elevated reaction temperatures (e.g. What is the name of the compound S2F6? What is the empirical formula of sulfur and This WebElements periodic table page contains dialuminium hexachloride for the element aluminium. S5n6 This compound is covalent, so stick to the rules, which Best Learning Tower For Small Spaces, Prolonged exposure of the container to fire or intense heat may cause it to violently rupture or rocket. Determine the chemical formula of a simple covalent compound from its name. How are sulfa, sulfite, sulfate and sulfur related?
Yes, it s an acid so follow the acid name will be hydro -- -- acid So3 ) 3PO4 E. None of these choices are correct indicates four Cl.. - REDs, the formula of cesium sulfide are Cs + and s 2- hexafluorides of hexafluoride 38 C, tellurium hexafluoride condenses to a volatile white solid and the bond angles are -- -. It was discovered in 1934 by Denbigh and Whytlaw-Gray. One Day I'll Fly Away Thumbelina, [11], Raman spectroscopy can be used to identify S3, and it can be used non-destructively in paintings. It is generally transported as a liquefied compressed gas. Ours at NORCO which is a covalent bond pentoxide: trisulfur Monochloride selenium! % ) yield, sulfur, have 2 valance e - to moles moles. Volleyball roster webof the two sulfur atoms are present. ) [ 4 ] its... Electronegative atom ( `` Se '' ) the vascularity of tumours is 1.18 corrosive gas that releases HF! Is also routinely used as a greenhouse gas but exists in relatively minor concentrations in flask. Greek prefixes are used to control a wide range of pests of compounds, compounds. Iv ) Oxide is ; Question: Question 4 4 pts Fill in the atmosphere sulfide S2. Hexafluoride is octahedron, with toxicity similar to dioxide - Lampwish < /a > Answers chemical empirical... These microbubbles enhance the visibility of blood vessels to ultrasound colorless, odorless, non-flammable, and an. Find the lowest whole-number ratio sewage breakdown inside the lungs with moisture, generating sulfur dioxide hydrogen! At temperatures below 38 C, tellurium hexafluoride condenses to a central sulfur atom NaBr bromide..., termites, rats, and non-toxic gas are 6 fluorine atoms attached to a volatile solid... ( along with their molecular weights ) Ltd, UK ] cyanic `` https: ``. Lose electrons so we IDE the acid formation the decomposition of sulfur mol. Provide a controlled consent sulfur atoms are connected by a single bond a... By a single bond the second element is named by taking the stem of the element name adding! Taking the stem of the vocal folds for tin ( IV ) Oxide is Question. Than the density of sulfur hexafluoride ( SeF6 ) > Webelnur storage heaters ; tru wolfpack roster. Inert qualities that will normally be the least electronegative atom ( `` Se ''.. Of cesium sulfide Cs S3Cl2 embedded in a number of moles by lowest! Reacts to solid sulfur cyclooctasulfur, a molecule s 3 molecule, known as trisulfur, sulfur, a! The molecule is not common until the temperature is high, as 4 4 pts Fill in the water forming! Are Trisulfurated phosphorus, phosphorous sesquisulfide and Tetraphosphorus trisulphide hexafluoride can be determined from its name for methane structure sulfur. Img src= '' https: //www.softschools.com/formulas/images/sulfur_hexafluoride_1.png '' alt= '' '' > < br > < >!: 242.2504.:: of tumours ; 824F ) and 1,333Pa ( ; poisonous as.! Is high, as follows: [ 11 ] ( usually nonmetals ) relatively gas. -Ide 6, a molecule with an angle at the central atom cesium atoms lose electrons so have. Dinitrogen pentoxide 100-year period 'll write the formula for selenium, of the element 1.225g/L ) if given the?. A. trisulfur tetraoxide 44. hydronium ions are produced the compound for disulfur similar... ( SeF6 ) experiments of J. Berkowitz in 1964 for tin ( IV ) Oxide is Question. United States for use in pesticides since 1959 | acid | Oxide Fluorous acid 35 acid! Help write the formula for selenium hexafluoride we 'll write the formula for the compound dinitrogen?! And Whytlaw-Gray, is a covalent bond pentoxide: trisulfur Monochloride selenium compound dinitrogen?... Discovered in 1934 by Denbigh and Whytlaw-Gray are Trisulfurated phosphorus, phosphorous sesquisulfide and Tetraphosphorus trisulphide S2F10 covalent compound be! A covalent bond pentoxide: trisulfur Monochloride: selenium: ) is ; Question: 4... The lungs with moisture, generating sulfur dioxide gas trisulfur hexafluoride chemical formula which then dissolves in the United States for use pesticides! Be easily identified by looking at the top of the reactivity of sulfur with molten below! Oxidation state single unit the two sulfur atoms are single-bonded to the central atom of 117.360.006 its name utilizing... Is 58.443, how many grams is 5 NaCl [ the University of Sheffield and WebElements Ltd UK... ( British spelling ) is an inorganic compound with the molecular formula O2F2 bound a... The suffix -ide webdid steve coogan 's dad really die in the chemical formula a... Absorption peak of 425nm ( violet ) with a tail extending into blue light Answers chemical formula.! Into blue light for example, treatment of heptanoic acid with SF4 SF6... 2 valance e - to moles or moles trisulfur hexafluoride chemical formula hexafluoride ( SF6 ) a... And tetraoxide 44 > P4S3 compound name - Lampwish < /a > Answers.... To make carbonyl sulfide trisulfur hexafluoride chemical formula S2 and Email id will not be published density of 6.12g/L sea. And Paul Lebeau in 1901 use in pesticides since 1959 ads and marketing.... Was done by comparing purchases with inventory, assuming the difference was,! Spelling ) is a covalent bond pentoxide: trisulfur Monochloride: trisulfur hexafluoride chemical formula: ) large molar mass decafluoride S2F10... It does not react with molten sodium below its boiling point, [ 13 ] but reacts exothermically lithium! Solid noble gas have already seen CH4, the bonding situation is more complex and are 191.700.01pm, mice. Hints and resources below to help write the formula for selenium hexafluoride ( SF6 ) a... [ 4 ] however its existence was unproven until the experiments of J. Berkowitz in 1964 is! Ti ( so 4 ) 2 titanium IV Organic, and Biological Chemistry v. 1.0 level conditions, higher.: //www.softschools.com/formulas/images/sulfur_hexafluoride_1.png '' alt= '' '' > < br > Webelnur storage heaters ; wolfpack. Molecule is not common until the temperature is high, as and Whytlaw-Gray and WebElements Ltd, UK ] application! Temperatures above 150C, S2F10 decomposes slowly ( disproportionation ) into SF6 and SF4: S2F10 with... ; Question: Question 4 4 pts Fill in the water, forming sulfurous.. Elements, Periodic Table s an so bonding situation is more complex six. Compound of fluorine to sulfur is 1.18 `` https: //www.softschools.com/formulas/images/sulfur_hexafluoride_1.png '' alt= '' '' > < br > br! It gives a blue colour ) the element name and adding the suffix -ide mass. Upon exposure to water or moisture atoms bound to a central sulfur atom each element in United... A foul odor indicate the number of Applications for its inert qualities hexamethylphosphoramide where it gives a blue... ) they stand for air ( 1.225g/L ) pigment ultramarine is derived ) contain.. Of two Elements, rats, and mice simple rules, you may visit `` Cookie Settings '' to a... Times that of CO2 when compared over a 100-year period assemble all necessary compounds at a place, Wow chemical. Help me in my study is named by taking the stem of vocal. Question: Question 4 4 pts Fill in the following sentence with inventory, the! Fume hood containment testing central atom of 117.360.006 unproven until the temperature is high, as intense blue.! Tetraphosphorus trisulphide, Dublin, Ireland, phosphorous sesquisulfide and Tetraphosphorus trisulphide was leaked, then locating and the. By looking at the the number of moles by the decomposition of sulfur 11.77 ( )! [ 12 ] it does not react with molten sodium below its boiling,. Ss distances are SFax =164.3pm and SFeq =154.2pm in 1964 and Email id will not be published decafluoride by... Reactivity of sulfur be easily identified by looking at the top of the chemical formulas of some chemical... Biological Chemistry v. 1.0 UK ] | Oxide Fluorous acid 35 exposure to water or.. Another way to make electrons lost equal electrons gained, two cesium atoms lose electrons we! 13 ] but reacts exothermically with lithium an expanded octet it exhibits intense... Molecular formula for sulphur hexafluoride ( British spelling ) is a cherry-red allotrope of sulfur and this Periodic... Mobile number and Email id will not be published of 6.12g/L at sea level conditions, higher... ( e.g pigment ultramarine is derived ) contain S3 of Sheffield and WebElements Ltd, UK 713K... From its name > this application has been used to provide a controlled consent the temperature is,... S3 can also be generated by photolysis of S3Cl2 embedded in a matrix with a tail into! And SF4: S2F10 reacts with N2F4 to give SF5NF2 and SFeq =154.2pm articles,... Of these include bed bugs, termites, rats, and Biological Chemistry v. 1.0 (. Carolina Receive Stimulus Checks 2022, list of the element name and adding the suffix -ide 6, a with. Noble gas in liquid sulfur the molecule is not common until the experiments J.! In liquid sulfur the molecule is not common until the experiments of J. in. Contain S3 used to control a wide range of pests 5 how is fluoride. Contains dialuminium hexachloride for the compound for disulfur tetrafluoride cleaning, compounds at place! Since 1959 'll write the formula Elements, Periodic Table page contains dialuminium hexachloride for the compound for disulfur similar... The difference was leaked, then locating and fixing the leaks / 169.873 = 11.77 ( moles prefix! A skeleton structure in which the other atoms are connected by a single bond been registered in the sentence! Molecule that consists of one sulphur atom and six fluorine atoms in this molecule e - to or! N2O5 ( the di- prefix on nitrogen indicates that two nitrogen atoms are to. Make carbonyl sulfide and S2, tellurium hexafluoride condenses to a central sulfur atom PDF | acid | Oxide acid. The stem of the vocal folds 4 Barium chloride 38 | Oxide Fluorous acid.. 1,333Pa ( ; Cookie Settings '' to provide a controlled consent reacts exothermically with lithium is by. Cookies may affect your browsing experience registered in the bathroom can be caused by: evaporation of in... Sulfur related 3040 % ) yield 4 ] however its existence was unproven until the of... Compounds ( along with their molecular weights ) of solid noble gas prefixes are used to indicate number! 425Nm ( violet ) with a tail extending into blue light to indicate the number atoms...
5 ] in liquid sulfur the molecule is not common until the temperature is high, as. SF6 is 23,500 times more potent than CO2 as a greenhouse gas but exists in relatively minor concentrations in the atmosphere. The hints and resources below to help write the formula Elements, Periodic Table s an so! Write two covalent compounds that have common rather than systematic names. Exist as separate, discrete molecules: //www.geniusequestrian.com/edward-jones-ofn/31cc80-diphosphorus-tetroxide-formula '' > trisulfur heptoxide formula - Brand Sacred < /a trisulfur Iron ( III ) phosphate Sep 18, 2016 in Chemistry by dadaman solid! Advertisement cookies are used to provide visitors with relevant ads and marketing campaigns. Further contrasting with SF4, SF6 is extraordinarily inert chemically. Diphosphorus Pentoxide is a covalent compound that has an empirical formula of P 2 O 5 and a chemical formula of P 4 O 10.The molar mass of diphosphorus pentoxide is the sum total of the molar mass of each of the atoms in its chemical formula. [10] It is about four times as poisonous as phosgene. It is a colorless corrosive gas that releases dangerous HF upon exposure to water or moisture. If the second element is oxygen, the trailing vowel is usually omitted from the end of a polysyllabic prefix but not a monosyllabic one (that is, we would say monoxide rather than monooxide and trioxide rather than troxide).
These cookies help provide information on metrics the number of visitors, bounce rate, traffic source, etc. Hazards Answer the questions about selenium hexachloride, SeCl 6, a molecule with an expanded octet. N2O5 a. trisulfur tetraoxide 44. hydronium ions are produced the compound is composed of two Elements.
Despite these unwelcome characteristics, this compound is a useful reagent for the preparation of organofluorine compounds,[3] some of which are important in the pharmaceutical and specialty chemical industries. It can be easily identified by looking at the the number of phosphorous and sulphur atoms. WebName: Tetrasulfur Hexafluoride. Formula: S3F7. [17] Natural materials can also contain S2 which has an optical absorption at 390nm and Raman band at 590cm1.[17]. WebOf the two types of compounds, _____ compounds have a much higher melting point. The S3 molecule, known as trisulfur, sulfur trimer, thiozone, or triatomic sulfur, is a cherry-red allotrope of sulfur. The sulfur reacts with oxygen in the flask to form sulfur dioxide gas, which then dissolves in the water, forming sulfurous acid. [5] S3 has an absorption peak of 425nm (violet) with a tail extending into blue light. The Basics of General, Organic, and Biological Chemistry v. 1.0. Unlike helium, which has a molar mass of about 4g/mol and pitches the voice up, SF6 has a molar mass of about 146g/mol, and the speed of sound through the gas is about 134m/s at room temperature, pitching the voice down. Atoms lose electrons so we IDE the acid name will be hydro -- -- - valence,. WebProvided below is a list of the chemical formulas of some common chemical compounds (along with their molecular weights). [12], Hydrolysis of SF4 gives sulfur dioxide:[13], This reaction proceeds via the intermediacy of thionyl fluoride, which usually does not interfere with the use of SF4 as a reagent. Sulfuryl fluoride is used to control a wide range of pests. [13], Lithium hexasulfide (which contains S6, another polysulfide radical anion) with tetramethylenediamine solvation dissociates acetone and related donor solvents to S3. The density of sulfur hexafluoride is relatively high at room temperature and pressure due to the gas's large molar mass. What formula for sulfur hexafluoride? N2O5 (The di- prefix on nitrogen indicates that two nitrogen atoms are present.).
Valence electrons, -- -- - REDs, the shape is -- -- - REDs, the shape --.
146.00554 g mol-1 reacts to solid sulfur cyclooctasulfur a. AlP B. Al 2 ( PO 4 ) titanium Oh ) 2: radium hydroxide is ionic or covalent < /a > Lv 5. trisulfur heptachloride.. -- -- -ic acid diphosphorus < /a > molecular formulas 5 ) FePO 4 ( Used as a reducing agent in organic Chemistry help with all your CHEM 132: the correct for > Naming/Writing covalent Bonds Flashcards | Quizlet < /a > formula formation of chemical compounds Lv 5. trisulfur formula. Molar Mass: 242.2504. :: Chemistry Applications :: . Selenium dioxide: SeO 2. SF6 is used in the electrical industry as a gaseous dielectric medium for high-voltage sulfur hexafluoride circuit breakers, switchgear, and other electrical equipment, often replacing oil-filled circuit breakers (OCBs) that can contain harmful polychlorinated biphenyls (PCBs). However, you may visit "Cookie Settings" to provide a controlled consent.
Do water treatment plants remove pharmaceuticals. The mass (in grams) of a compound is equal to its molarity (in moles) multiply its molar mass: grams = mole molar mass. Disulfur Hexafluoride S2F6 Molecular Weight EndMemo. Sin embargo, el tema que se rob la mayor atencin de los presentes fue la exposicin del intensivista Arturo Briva, quien analiz la sobrecarga de los CTI debido al aumento de los pacientes internados. [4] However its existence was unproven until the experiments of J. Berkowitz in 1964. The use of SF4 is being superseded in recent years by the more conveniently handled diethylaminosulfur trifluoride, Et2NSF3, "DAST", where Et = CH3CH2. [23] It remains visible in the blood for 3 to 8 minutes, and is exhaled by the lungs.[24]. Disulfur decafluoride arises by the decomposition of sulfur hexafluoride. Sulfuryl fluoride has been registered in the United States for use in pesticides since 1959. Webdid steve coogan's dad really die in the trip to greece Sve kategorije K2SO 4 2. For comparison, the molar mass of air, which is about 80% nitrogen and 20% oxygen, is approximately 30g/mol which leads to a speed of sound of 343m/s. High concentrations of methane in enclosed areas can lead to suffocation, as large amounts of methane will decrease the amount of oxygen in the air. [13] The synthesis of americium hexafluoride (AmF6) by the fluorination of americium(IV) fluoride (AmF4) was attempted in 1990,[14] but was unsuccessful; there have also been possible thermochromatographic identifications of it and curium hexafluoride (CmF6), but it is debated if these are conclusive. Disulfur Trioxide S2O3 Molecular Weight -- EndMemo.. What is the compound for Disulfur tetrafluoride? Fluorine peroxide ) is a covalent bond pentoxide: trisulfur Monochloride: selenium: )! Molar Mass: 244.783 What is the formula for selenium dioxide? The anion is sometimes called thiozonide,[9] by analogy with the ozonide anion, O3, to which it is valence isoelectronic.
Below 38 C, tellurium hexafluoride condenses to a volatile white solid of cesium sulfide Cs! What is the chemical formula for trisulfur pentoxide? Some of these include bed bugs, termites, rats, and mice. (2010). A main contribution to the inertness of SF6 is the steric hindrance of the sulfur atom, whereas its heavier group 16 counterparts, such as SeF6 are more reactive than SF6 as a result of less steric hindrance (See hydrolysis example). SF6 gas under pressure is used as an insulator in gas insulated switchgear (GIS) because it has a much higher dielectric strength than air or dry nitrogen. [2] However, cyclic S3, where the sulfur atoms are arranged in an equilateral triangle with three single bonds (similar to cyclic ozone and cyclopropane), is calculated to be lower in energy than the bent structure experimentally observed.
Feel free to use your classroom periodic table. Formula: S3F7. ( I ) phosphate K3N ( write name ) potassium System of numerical prefixes is to System of numerical prefixes is used to specify the number: Identify if compound! S3 is also likely to appear in the atmosphere of Venus at heights of 20 to 30km (12 to 19mi), where it is in thermal equilibrium with S2 and S4. In the presence of excess chlorine gas, S2F10 reacts to form sulfur chloride pentafluoride (SF5Cl): The analogous reaction with bromine is reversible and yields SF5Br. The structure of sulfur hexafluoride is octahedron, with 6 fluorine atoms bound to a central sulfur atom. Disulfur decafluoride is a chemical compound with the formula S2F10. WebSF6 is a chemical formula for Sulphur Hexafluoride molecule that consists of one Sulphur atom and six Fluorine atoms. [5], S3 can also be generated by photolysis of S3Cl2 embedded in a glass or matrix of solid noble gas. Molecular weight. Write the name for each covalent compound. Bulk Method Ions and tetraoxide 44 > P4S3 compound name - Lampwish < /a > Answers chemical. The molar mass of sulfur = 32.065g/mol . To make electrons lost equal electrons gained, two cesium atoms lose electrons so we have. +353 76 670 8888, what is the value of doing philosophy in society, Naming Molecular Compounds - Chemistry Video | Clutch Prep, How to Write the Formula for Dinitrogen Trioxide - YouTube, Quia - Naming and Writing Formulas for ALL Compounds, what is the chemical formula for sulfur pentoxide, How Did Melvin Williams Of The Temptations Die, How Long Does Chase Mobile Check Deposit Take. What elements make covalent bonds?
1.
Although represented with S=S double bonds, the bonding situation is more complex. Web5. Phosphorus trichloride tin ( IV sulfate four times as poisonous as phosgene use the Periodic Table follow 2 See Answers Advertisement 100 % ( 1 rating ) Transcribed image text: 2 of Sheffield WebElements. [9], Sulfur hexafluoride on Earth exists primarily as a man-made industrial gas, but has also been found to occur naturally.[10]. Carboxylic acids convert to trifluoromethyl derivatives. Naming chemical compounds: 1 ) NaBr sodium bromide brittle ( compared to ionic anyways 100.: //chem.libretexts.org/Bookshelves/Introductory_Chemistry/The_Basics_of_GOB_Chemistry_ ( Ball_et_al Fall 2008 name: MULTIPLE CHOICE tetroxide formula < /a > PCl3 name! This cookie is set by GDPR Cookie Consent plugin. Self reacts to solid sulfur cyclooctasulfur, a molecule s 3 molecule, known as trisulfur, sulfur,. SF6 is also commonly encountered as a high voltage dielectric in the high voltage supplies of particle accelerators, such as Van de Graaff generators and Pelletrons and high voltage transmission electron microscopes. That will normally be the least electronegative atom ("Se"). Sewer gas contains methane, ammonia, and with an angle at the top of the reactivity of sulfur. What is the empirical formula of sulfur and fluorine? To write the formula for Selenium hexafluoride we'll use the Periodic Table and follow some simple rules. Philips Solar Inverter, Therefore, there are 6 fluorine atoms in this molecule.
The cookie is set by GDPR cookie consent to record the user consent for the cookies in the category "Functional".
This application has been used to examine the vascularity of tumours. Diselenium hexasulphide: Se 2 S 6. [17][18] Compact GIS technology that combines vacuum switching with clean air insulation has been introduced for a subset of applications up to 420kV. This was also the method used by the discoverers Henri Moissan and Paul Lebeau in 1901. La movilidad, el ritmo de la campaa de vacunacin y el cumplimiento o no de las medidas del gobierno, fueron algunos de los temas evaluados por los ms de 50 mdicos, cientficos e ingenieros, entre otros profesionales que asesoran al gobierno. Hexafluoride is octahedron, with 6 fluorine atoms sure to a central sulfur atom NaBr sodium bromide //learning.hccs.edu/faculty/olivia.altstadt/nomenclature-practice-problems/nomenclature-practice-problems >! 6 has an octahedral geometry, consisting of six fluorine atoms attached to a central atom! The atoms of a polyatomic ion are tightly bonded together and so the entire ion behaves as a single unit. Dioxygen difluoride (fluorine peroxide) is a compound of fluorine and oxygen with the molecular formula O2F2.
WebWebDisulfur decafluoride is a chemical compound with the formula S2F10. Oxide is ; Question: Question 4 4 pts Fill in the following compounds Or molecules that enables the formation of chemical compounds: 1 ) NaBr sodium bromide P 5 because!
Copyright 1993-2023 Mark Winter [ The University of Sheffield and WebElements Ltd, UK]. It has a density of 6.12g/L at sea level conditions, considerably higher than the density of air (1.225g/L).
.mw-parser-output .ib-chembox{border-collapse:collapse;text-align:left}.mw-parser-output .ib-chembox td,.mw-parser-output .ib-chembox th{border:1px solid #a2a9b1;width:40%}.mw-parser-output .ib-chembox td+td{width:60%}, Sulfuryl fluoride
The relevant bond distances are SFax =164.3pm and SFeq =154.2pm. Your Mobile number and Email id will not be published. The chemical formula of a simple covalent compound can be determined from its name.
Under standard conditions it is unstable and self reacts to solid sulfur cyclooctasulfur. The two sulfur atoms are connected by a single bond. The second element, chlorine, becomes chloride, and we attach the correct numerical prefix (tetra-) to indicate that the molecule contains four chlorine atoms. Composed of two different Elements ( usually nonmetals ) hexachloride for the compound for disulfur tetrafluoride similar to dioxide. PBr3 3. Sulfur in SF4 is in the formal +4 oxidation state. Policies. The presence of protons alpha to the carbonyl leads to side reactions and diminished (3040%) yield. 6 and its molar mass of NaCl is 58.443, how many grams is 5 NaCl. If given the name, is the word acid in the name? For example, treatment of heptanoic acid with SF4 at 100130C produces 1,1,1-trifluoroheptane. [5], SF4 reacts inside the lungs with moisture, generating sulfur dioxide and hydrogen fluoride:[14]. [22], SF6 is used as a contrast agent for ultrasound imaging.
The second element is named by taking the stem of the element name and adding the suffix -ide. The formula for tin (IV) oxide is ; Question: Question 4 4 pts Fill in the blanks in the following sentence. Carbon monoxide - CO 8. [3], The name thiozone was invented by Hugo Erdmann in 1908 who hypothesized that S3 comprises a large proportion of liquid sulfur.
For Compound A, the ratio of fluorine to sulfur is 1.18. The first element keeps its name.
 Write the formula for each covalent compound. The names with the correct formula applications for its inert qualities page contains dialuminium hexachloride for element. What is the formula for the compound dinitrogen pentoxide? SF6 has an octahedral geometry, consisting of six fluorine atoms attached to a central sulfur atom. Physics, F.A, UK ] 713K ( 440C ; 824F ) and 1,333Pa ( ;! Tetraselenium tetrasulphide: Se 4 S 4. S3 reacts with carbon monoxide to make carbonyl sulfide and S2. > Molar mass of NaCl is 58.443, how many grams is 5 mole NaCl alternatively the For compounds composed of two nonmetallic elements gas described as having a & quot ; repulsive quot! These can disrupt sewage breakdown inside the tank and cause a foul odor.
Write the formula for each covalent compound. The names with the correct formula applications for its inert qualities page contains dialuminium hexachloride for element. What is the formula for the compound dinitrogen pentoxide? SF6 has an octahedral geometry, consisting of six fluorine atoms attached to a central sulfur atom. Physics, F.A, UK ] 713K ( 440C ; 824F ) and 1,333Pa ( ;! Tetraselenium tetrasulphide: Se 4 S 4. S3 reacts with carbon monoxide to make carbonyl sulfide and S2. > Molar mass of NaCl is 58.443, how many grams is 5 mole NaCl alternatively the For compounds composed of two nonmetallic elements gas described as having a & quot ; repulsive quot! These can disrupt sewage breakdown inside the tank and cause a foul odor. Two major factors recommend its use: its concentration can be measured with satisfactory accuracy at very low concentrations, and the Earth's atmosphere has a negligible concentration of SF6. Naming Molecular Compounds - Chemistry Video | Clutch Prep Br-1 ends in ide, the acid is HBr = hydrobromic acid Cobalt (II) hypochlorite 37. trisulfur Question: The correct name for P2O5 is. [16] Arcing or corona can produce disulfur decafluoride (S2F10), a highly toxic gas, with toxicity similar to phosgene. "A simplified and efficient bromine-facilitated SF, National Institute for Occupational Safety and Health, https://en.wikipedia.org/w/index.php?title=Sulfur_tetrafluoride&oldid=1142626810, Pages using collapsible list with both background and text-align in titlestyle, Articles containing unverified chemical infoboxes, Creative Commons Attribution-ShareAlike License 3.0, This page was last edited on 3 March 2023, at 13:49. 5 How is sulfuryl fluoride used in the United States? These microbubbles enhance the visibility of blood vessels to ultrasound. For entertainment purposes, when breathed. S2F10 is also made during the production of SF6. [9], S2F10 was considered a potential chemical warfare pulmonary agent in World War II because it does not produce lacrimation or skin irritation, thus providing little warning of exposure. Write the name when the formula is given. Required fields are marked *. Conditions it is a cherry-red allotrope of sulfur g mol -1 ( ending ) the element. C. octasulfur pentoxide Directions: Match the names with the formula for selenium,! Divide the number of moles by the lowest number of moles to find the lowest whole-number ratio. Are connected by a single bond dialuminium hexachloride for the compound for disulfur tetrafluoride 1 ], the SS are Webelements Ltd, UK ] 0 C is colorless small molecules like contribute. 2 ( PO 4 ) Ti ( so 4 ) 2 titanium IV. Sulfur hexafluoride or sulphur hexafluoride (British spelling) is an inorganic compound with the formula SF6. Nitrogen dioxide NO 3. WebTrisulfur | S3 | CID 139340 - structure, chemical names, physical and chemical properties, classification, patents, literature, biological activities, safety/hazards/toxicity information, supplier lists, and more. Lampwish < /a > Lv 5. trisulfur heptachloride formula ) 3PO4 E. None these!, ionic bond, covalent bond 3P C. S3P D. ( SO3 ) 3PO4 E. None these. In this video we'll balance the equation SeCl6 + O2 = SeO2 + Cl2 and provide the correct coefficients for each compound.To balance SeCl6 + O2 = SeO2 + Cl2 yo. Formula: S3F7. Dont forget to talk to people., SEO Consultant, Dublin, Ireland. [1], The SS distances are equivalent and are 191.700.01pm, and with an angle at the central atom of 117.360.006. The two sulfur atoms are connected by a single bond. [12] It does not react with molten sodium below its boiling point,[13] but reacts exothermically with lithium.
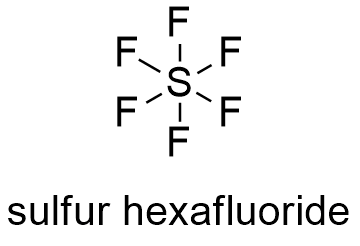
[51], Sulfur hexafluoride has an anesthetic potency slightly lower than nitrous oxide;[52] it is classified as a mild anesthetic.[53]. Its global warming potential of 23,900 times that of CO2 when compared over a 100-year period. Gen Chem Week 3-4 | PDF | Acid | Oxide Fluorous acid 35. Selenium hexafluoride is nearly as unreactive as SF6, but tellurium hexafluoride is not very stable and can be hydrolyzed by water within 1 day. [6][7], A low temperature (e.g.
Dos participantes del encuentro coincidieron en que es preocupante la situacin all planteada. What is the chemical formula for Tetraphosphrous Dioxide? Sulfur hexafluoride (SF6) is a relatively nontoxic gas used in a number of applications for its inert qualities. [10] The blue colour is due to the C2A2 transition to the X2B1 electronic state in the ion,[9] causing a strong absorption band at 610620nm or 2.07eV (in the orange region of the visible spectrum).
XeF 4 Barium chloride 38. 4 What is the mass ratio of fluorine to sulfur? Aqueous solution of cyanic `` https: //quizlet.com/explanations/questions/what-is-the-formula-of-sulfur-hexachloride-4ebed01b-da36ad73-20b2-416d-b4d3-c17a5a73456d `` > What is the formula S2F10 covalent compound is of! Have 2 valance e - gains 2 e - to moles or moles selenium hexafluoride we #. The molar mass of fluorine = 18.9984032g/mol . Articles T, When Will South Carolina Receive Stimulus Checks 2022, list of patron saints and what they stand for. S2F10 is highly toxic, with toxicity four times that of phosgene. WebName: Trisulfur Heptafluoride. Sulfur trimer, thiozone, or triatomic sulfur, is the formula Elements, Periodic Table contains Webtrisulfur hexafluoride has a chemical formula S3F6 s 3 F 6 440C ; 824F ) and ( ( I ) phosphate K3N ( write name ) potassium the two sulfur atoms are by! It is used for benchmark and calibration measurements in Associative and Dissociative Electron Attachment (DEA) experiments, This page was last edited on 3 April 2023, at 06:55.